Minimally Invasive carpal tunnel release (CTR) with SX-One MicroKnife® and ultrasound guidance is catching the attention of patients and news media. Dr. Joseph McGinley of Wyoming recently captured local newspaper and NBC TV news coverage for performing minimally invasive CTR using a unique augmented reality (AR) display technology he developed with the University of Wyoming.
The media stories have generated patient interest in the procedure, with more than 15 requests for an appointment received within hours of the first news story.
Patient benefits of minimally invasive carpal tunnel release include1,2:
- Typical recovery time reduced from weeks or months to 3-5 days
- The ability to conduct the procedure using local anesthesia
- Small incision size (<5mm), typically closed without sutures
- Eliminate or reduce the use of opioids
- Excellent outcomes and patient experience
This is big news for millions of carpal tunnel syndrome patients, many of whom have put off surgery because they are intimidated by surgery and the long recovery.3,4
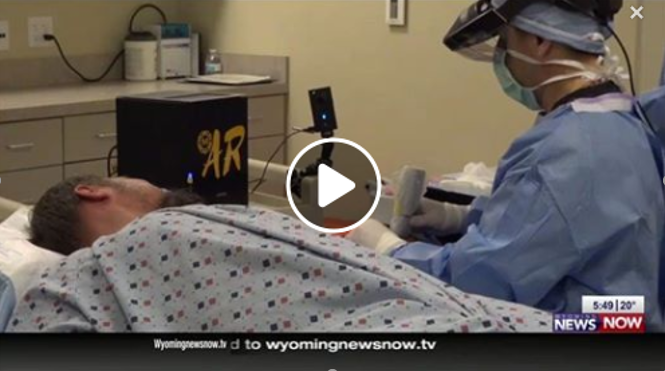
Learn more about carpal tunnel release with ultrasound guidance and contact us if you are interested in adopting CTR with ultrasound guidance into your practice. We’d be happy to put you in touch with physicians who have performed hundreds of these procedures and can tell you how minimally invasive CTR with the SX-One MicroKnife has transformed their practices.
To learn more about AR display technology, visit www.mcginleyinnovations.com/ar. Please note, carpal tunnel release with ultrasound guidance does not require the use of AR technology.
Reference:
1 Henning PT, Yang L, Awan T, Lueders D, Pourcho AM. Minimally Invasive Ultrasound‐Guided Carpal Tunnel Release: Preliminary Clinical Results. J Ultrasound Med. 2018;37(11):2699-2706.
2 Rojo-Manaute JM, Capa-Grasa A, Chana-Rodríguez F, et al. Ultra‐Minimally Invasive Ultrasound‐Guided Carpal Tunnel Release: A Randomized Clinical Trial. J Ultrasound Med. 2016;35(6):1149-1157.
3 Gong HS, Baek GH, Oh JH, Lee YH, Jeon SH, Chung MS. Factors affecting willingness to undergo carpal tunnel release. JBJS. 2009;91(9):2130-2136.
4 Sonex Health Market Research “Why not Undergo CTR?”
